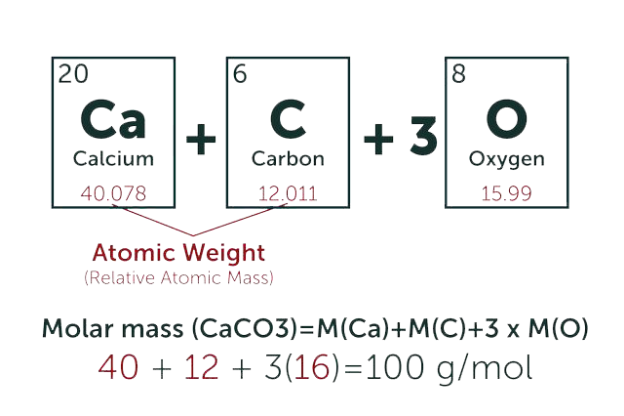The molecular weight also known as the molecular mass is the sum of the weight of the atoms to be measured i.e. the total weight of each the atomic masses of the atoms being considered. The unit is given as AMU or u. Or gram per mole (g/mol)
In calculating molecular weight manually, it is important to know the following;
First, that the mole as used in chemistry is the standard method to communicate the amount of substance present in the molecule and is different from the molecule. It is a unit of all that is present in a sub substance a mole of anything has 6.022 x 1023 molecules. Next is the relative atomic masses of the element that are to be measured. While the molecule is the particle that represents the molecular compound and the diatomic element.
Calculating for molecular weight manually takes 4 easy steps;
- Write out the elements and the number of the present in the chemical substance.
- Determine the atomic weight of each of the elements present in the substance (with the aid of the periodic table)
- Multiply the number of atoms present with their respective atomic weight
- After multiplying, sum up the values, the result becomes the molecular weight for that chemical substance for example when calculating the molecular mass of water, whose chemical formula is H2O.

There are three molecules present in it consisting of two hydrogen atoms and one oxygen atom. Consulting the periodic table the masses are one (1) for hydrogen and sixteen (16) for oxygen. Calculating you have 2(1) + 1 (16) = 18. So, therefore, the molecular mass of water (H2o) can be given as 18g/mol. As observed, the subscript number attached to the atom is multiplied by the atomic masses of the given atom.
Another important information to note there is a distinction between molar mass and the other concept which is the molecular weight. Both should not be confused for one another for they have different significance, the molar weight of a substance is the mass (usually depicted with a mole in mind) in that substance where the mole can be the number of ions, atoms, molecules, and electrons in an element. Whereas the other has to do with the mass which belongs to a single molecule in that substance mixing these two up can cause parameter error
Using the molecular weight calculator online, you just need to be able to write out the equation of the molecule and input it in the space provided, there, the value pops up immediately. If you type H2O into the bar it gives you 18.0152 g/mol. The extra decimal comes up because of the high accuracy of the calculator.
Molecular weight calculator online is easy to use since there is already a database of the periodic table that is used whenever it is needed, so therefore it leads to lesser error when calculating for your molecular mass. What is only needed is to be able to write out the chemical equation and identify the multipliers. When using a molecular calculator, the formulas can only have 2 brackets and the crystallization molecule should be placed last.
The molecular weight calculator can either be accessed online or installed on your desktop.